Carbohydrates
- Know the difference between monosaccharides, disaccharides and polysaccharides.
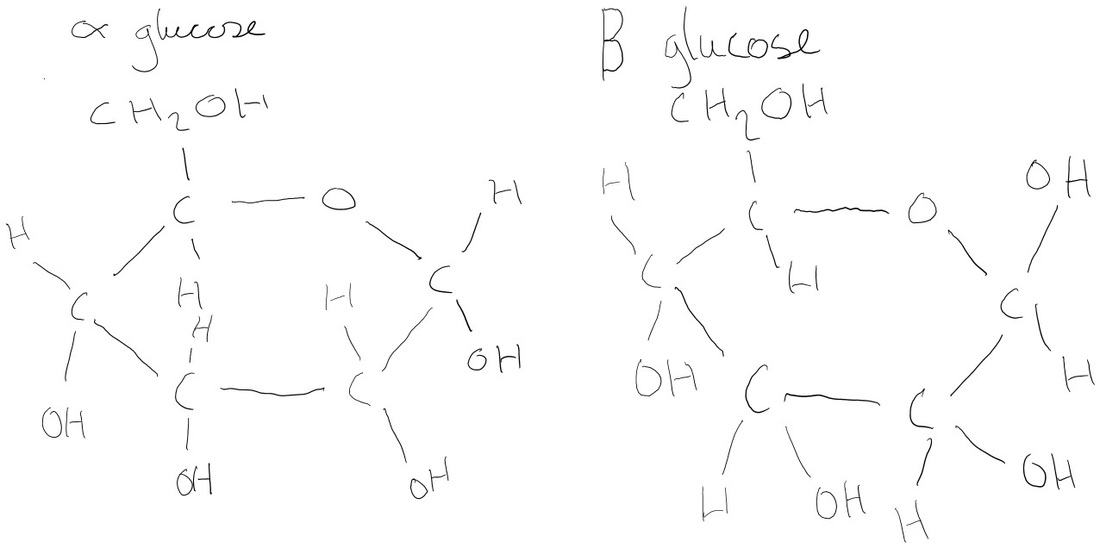
- Know the struture of the hexose glucose (alpha and beta) and the pentose ribose.
- Understand how the monosaccharides (glucose, fructose, galactose) join to form disaccharides (sucrose, lactose and moltose) and polysaccharides (starch formed from amylose and amylopectin; glycogen) through condensation reactions forming glycosidic bonds, and how these can be split through hydrolysis reactions.
- Understand how the structure of glucose, starch. glycogen and cellulose relates to their function.
Carbohydrates
Monosaccharides – simple sugars in which there are one oxygen atom and two hydrogen atoms, triose (n = 3), pentose (n = 5), hexose (n = 6) where n is the carbon atoms. Glucose is an isomer as there is alpha and beta. Beta the hydroxide is pointing in the direction of the 6 carbon.
Disaccharides – are made up of two monosaccharides joined together. They join in a condensation reaction and a molecule of water is removed, the bond between two simple sugars is known as a glycosidic bond.
If carbon 1 and 4 are bonded it is a 1, 4 glycosidic bond
If carbon 1 and 6 are bonded it is a 1, 6 glycosidic bond
Sucrose
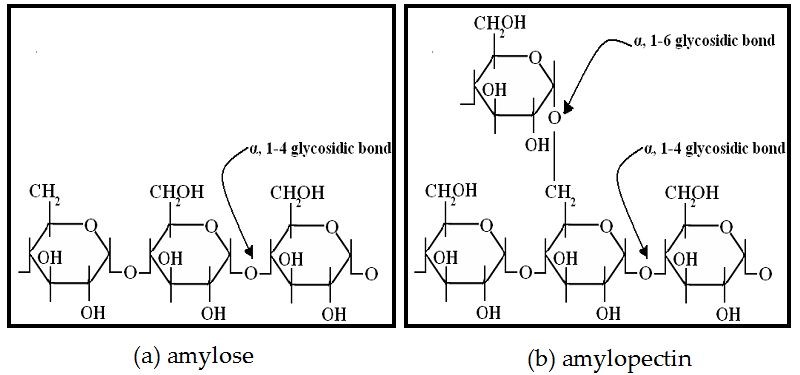
Starch
Amylose
Monosaccharides – simple sugars in which there are one oxygen atom and two hydrogen atoms, triose (n = 3), pentose (n = 5), hexose (n = 6) where n is the carbon atoms. Glucose is an isomer as there is alpha and beta. Beta the hydroxide is pointing in the direction of the 6 carbon.
Disaccharides – are made up of two monosaccharides joined together. They join in a condensation reaction and a molecule of water is removed, the bond between two simple sugars is known as a glycosidic bond.
If carbon 1 and 4 are bonded it is a 1, 4 glycosidic bond
If carbon 1 and 6 are bonded it is a 1, 6 glycosidic bond
Sucrose
- Stored in plants
- Alpha glucose + alpha fructose
- Sugar / main carbohydrate found in milk
- Alpha glucose + beta galactose
- Malt sugar, found in germinating seeds
- Alpha glucose + alpha glucose
Starch
Amylose
- An unbranched polymer made up of between 200 and 5000 glucose molecules
- The molecule spirals which makes it more compact for storage
- Made up of purely 1, 4 glycosidic bonds
- Releases glucose slowly over a period
- A branched polymer of glucose molecules
- Glucose can easily be ripped off when energy is needed
- Made up of both 1, 4 glycosidic bonds and 1.6 glycosidic bonds
- Rapid, on demand glucose
Glycogen
Cellulose
- Only energy store found in animals
- Like amylopectin but more 1, 6 glycosidic bonds meaning more branching
- Can be broken down rapidly
- Ideal source of glucose for active tissues
Cellulose
- Important structural material in plants, the cell wall gives strength and support
- Insoluble
- Made up of beta glucose and held together by 1, 4 glycosidic bonds which are inverted
- Inversion causes hydrogen bonds between the OH- and H+ which stick out (crosslinking)
- Crosslinking = Strength
- Long and straight
- Cannot be digested by many animals
- Alpha glucose
- Both 1, 4 and 1, 6 glycosidic bonds
- Compact globular molecules that are good for storage
- Source of energy in the diet for many animals