Lipids
- Understand how a triglyceride is synthesised including formation of ester bonds during condensation reactions between glycerol and three fatty acids
- Know the difference between saturated and unsaturated lipids
- Understand how the structure of lipids relates to their role in energy storage , waterproofing and insulation. Understand how the structure and properties of phospholipids relate to their function in cell membranes
Lipids
Lipids are an organic chemical that are used for all cell membranes and are used as an energy store. Fats are chemically similar to oils but are solid at room temperature, oils are liquid at room temperature. Lipids are made up of Carbon, hydrogen and oxygen, they are also made up of fatty acids and glycerol. They are combined using ester bonds. Glycerol has the chemical formula C3H8O3
The structure of a fatty acids is constant. The fatty acids are made up of a long hydrocarbon chain – a back bone of carbon atoms with hydrogen attoms attached, and a carboxyl group (-C=O/-OH). The change in fatty acids comes from:
Properties of Lipids
Phospholipids are used in cell membranes.
Lipids are an organic chemical that are used for all cell membranes and are used as an energy store. Fats are chemically similar to oils but are solid at room temperature, oils are liquid at room temperature. Lipids are made up of Carbon, hydrogen and oxygen, they are also made up of fatty acids and glycerol. They are combined using ester bonds. Glycerol has the chemical formula C3H8O3
The structure of a fatty acids is constant. The fatty acids are made up of a long hydrocarbon chain – a back bone of carbon atoms with hydrogen attoms attached, and a carboxyl group (-C=O/-OH). The change in fatty acids comes from:
- The length of the carbon chain
- The length of the back bone carbon chain will affect physical and chemical properties of the molecule
- Saturated or unsaturated
- If the fatty acid is unsaturated it will have either one(monounsaturated) or more than one (polyunsaturated) double bond
Properties of Lipids
- High energy value
- Lipids contain many carbon hydrogen bonds, when these are broken carbon dioxide and water are the final products. They are oxidised in respiration and are three times more efficient at energy storage than carbohydrates
- Hydrophobic
- Oils are used in the fur / feathers of animals as waterproofing. This is because the lipid tail of molecule is hydrophobic as the water is polar. They are good insulators as they have thick fat layers that trap air inside.
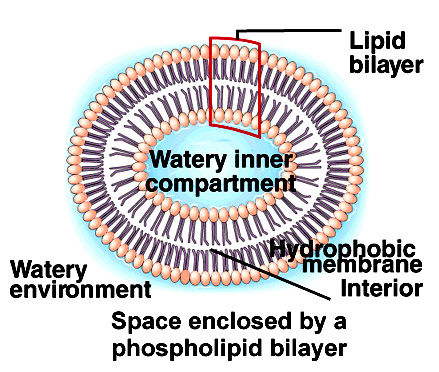
- Phospholipids have a similar structure to triglycerides, but with a phosphate group in place of one fatty acid chain.
- Phospholipids have a polar hydrophilic head (the negatively-charged phosphate group)
- Phospholipids have two hydrophobic tails (the fatty acid chains)
Phospholipids are used in cell membranes.
- When mixed with water, phospholipids form droplet spheres with a double-layered phospholipid bilayer.
- The hydrophilic dead faces the water with the tails facing one another. This traps a compartment of water at the centre. This causes trapped spheres of water as the surrounds are hydrophobic.
- This is called liposome, and is similar to a membrane surrounding a cell