Investigate how enzyme concentration affects the initial rate of an enzyme controlled reaction
This experiment was used to see how different concentrations of enzymes affected how quickly the reaction would normally occur. In this example we used trypsin at the concentrations 0.2%, 0.4%, 0.6%, 0.8% and 1% as a control / to calibrate the colorimeter.
Equipment Used:
1)Dilute the trypsin with distilled water, you will need to produce 0.2%, 0.4%, 0.6% and 0.8%. This is because concentration of enzyme is our independent variable, meaning we change it to see what effect it has on our experiment.
2)Calibrate the colorimeter, this is done by placing a cuvette full of clear liquid into the colorimeter and setting the colorimeter to 100%. This means that a fully clear liquid will be 100% of light passing through.
3)Add 2cm3 of trypsin to 2cm3 of milk suspension in a cuvette and add to colorimeter, this must be done quickly so that not too much of the reaction occurs before gathering results. When placed in colorimeter record the % light at regular intervals. For our experiment we used every 10 seconds for 5 minutes or until a constant result for sometime
4)Wash / replace cuvette and repeat for different concentrations, as we are investigating the effect of concentration we need to only be changing the concentration to be able to have reliable results.
Variables
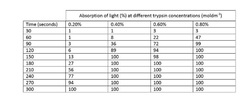
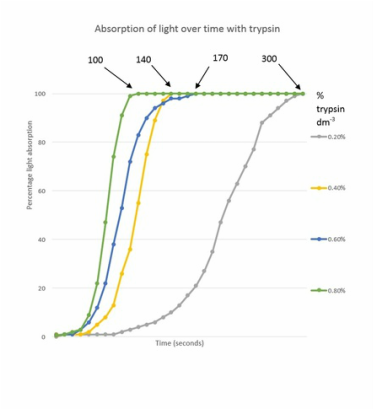
For our results we measured % of light passing through the solution at regular intervals of ten seconds. With this we can work out a value for the rate of reaction. It is important to use the initial rate of reaction as opposed to the average over a longer period of time as some of the reactions finished quickly, for example 0.8% trypsin fully reacted in 100 seconds but if we were to work out the rate of reaction over 300 seconds we would get a much lower value as this implies the reaction takes 300 seconds when actually many of them finished early.
This experiment was used to see how different concentrations of enzymes affected how quickly the reaction would normally occur. In this example we used trypsin at the concentrations 0.2%, 0.4%, 0.6%, 0.8% and 1% as a control / to calibrate the colorimeter.
Equipment Used:
- Skimmed milk powder suspension (2%)
- This was the protein that was being reacted, it is normally milky and using a colorimeter we can quantify the clarity of the liquid when reacted
- Standard protease (trypsin) solution (1%)
- This is our biological reactant with the milk, it is default 1% concentration but by mixing it with distilled water we can form our weaker concentrations to use
- Six test tubes and rack
- These were used to dilute our trypsin, for example a 0.2% solution from a 1% solution requires four parts water and one part trypsin
- 5cm3 pipettes
- Used for measuring the water and trypsin into the test tubes
- Eye protection, glasses
- Protease is an irritant as it digests protein so protection is mandatory
- Cuvettes
- A cuboid shape container which contains up to 4cm3 of liquid. It parallel sides it has opaque surfaces. This is used in placing the cuvette in the colorimeter
- Distilled water
- Distilled water is used to dilute the trypsin it has to be pure as any mineral ions or impurities would change the experiment slightly and would make it a poor medium for dilution
- Colorimeter
- A colorimeter shines a light from a bulb into a sensor which records the amount of light that reaches the sensor in relation to how much was projected from the bulb. This is used to quantify the liquids clarity as initially it is white and cloudy due to the milk but after the trypsin has had time to digest the protein it turns clear
1)Dilute the trypsin with distilled water, you will need to produce 0.2%, 0.4%, 0.6% and 0.8%. This is because concentration of enzyme is our independent variable, meaning we change it to see what effect it has on our experiment.
2)Calibrate the colorimeter, this is done by placing a cuvette full of clear liquid into the colorimeter and setting the colorimeter to 100%. This means that a fully clear liquid will be 100% of light passing through.
3)Add 2cm3 of trypsin to 2cm3 of milk suspension in a cuvette and add to colorimeter, this must be done quickly so that not too much of the reaction occurs before gathering results. When placed in colorimeter record the % light at regular intervals. For our experiment we used every 10 seconds for 5 minutes or until a constant result for sometime
4)Wash / replace cuvette and repeat for different concentrations, as we are investigating the effect of concentration we need to only be changing the concentration to be able to have reliable results.
Variables
- Independent Variable
- The concentration of trypsin in each experiment, we used 0.2%, 0.4%, 0.6% and 0.8% as it gave us multiple experiments whilst the difference in results was still quite large
- Dependant Variable
- The % light passing the cuvette and liquid which in turn means we are measuring the opacity or translucence of the liquid medium in which the reaction occurs. This is recorded on how this value changes over time, every 10 seconds
- Control
- Type of enzyme, volume of milk used, substrate, external conditions (pH and temperature). If we were to change the type of enzyme, volume of milk or used a different substrate our experiment would have been different as some enzymes work differently with different proteins and if we increased the milk volume the reaction would have taken longer. Enzymes also have an optimum pH and temperature and so if the volume was changed in any way it may make the reaction quicker or slower if it was closer or further away from the optimum.
For our results we measured % of light passing through the solution at regular intervals of ten seconds. With this we can work out a value for the rate of reaction. It is important to use the initial rate of reaction as opposed to the average over a longer period of time as some of the reactions finished quickly, for example 0.8% trypsin fully reacted in 100 seconds but if we were to work out the rate of reaction over 300 seconds we would get a much lower value as this implies the reaction takes 300 seconds when actually many of them finished early.
This table shows the results that another group had, although we recorded every 10 seconds for the purpose of this table I have used larger intervals. We can see, with the large intervals emphasising that the 0.4%, 0.6% and 0.8% all finished relatively close to one another and the 0.2% finished vastly slower. This could perhaps be because after a certain point adding more enzyme will not help as all the substrate will be reacted with the a set number of active sites and that adding more enzymes just saturates the liquid but does not help the rate. In the case of 0.2% the reason for the long time to react the substrate was because there were fewer enzymes for the same volume of substrate meaning the enzymes had to react with more proteins each then in any other experiment which inevitably takes longer.
Evaluation
In this experiment there were mnay things I could have done better to improve my reliability. These would help me get results that would be more reliable and maybe more accurate
- The quality of the cuvettes
- If my cuvette were to be scratched this would result in a greater absorbance of light. If it was my reference cuvette this would have made all my experiments could exceed 100% as 100%, in this case would be with a less than average amount light passing through (as more is absorbed) which cannot happen with a normal cuvette. If it was a cuvette used for a reaction, that particular reaction could never reach 100% as less light can pass through but 100% is set correctly by a normal cuvette. Before doing the experiments I should attempt to get the same brand of cuvette so the thickness of plastic and volume is constant and also to check for scratches and defects on my equipment and change the ones that would distort my readings
- Using a datalogger
- For my experiment I used a colorimeter which also needs a stop watch and a stop watch to be able to record the % displayed at a certain time. This means that when the stopwatch displays a ten second value the person has to react to this and call out that it has been reached. The person looking at the colorimeter has to react to it and record the value. This time in-between reaching the desired interval and the value given would never be constant each time so it cannot be adjusted for so easily. To remove this I would instead use a datalogger attached to a light detector and a light source. This gives the benefit of being able to look at your results at the end and the recording at given intervals is done automatically removing this human error which would have changed our data
- The point of combining the trypsin and milk
- For our experiment we could not have the cuvette in the colorimeter before we added the trypsin and then placed it in the colorimeter. In this time some of the protein would have started reaction so the recordings are all inconsistently a second or so behind. To stop this we could try place the trypsin into the cuvette when it is in the colorimeter