Objective
Safety
Variables
Equipment
Method
- Know how to carry out an investigation to determine the osmotic potential and therefore water potential of plant epidermal cells
- Osmosis is the net movement of water particles from an area of low water potential to an area of high water potential through a partially permeable membrane
- Incipient plasmolysis is when the cell membrane begins to pull away from the cell wall as the protoplasm shrinks
Safety
- Take care with glassware, mounting needles and cutting equipment. If glass cracks / smashes it becomes very sharp
Variables
- Independent
- Concentration of salt solution (mol dm-3‑)
- Dependant
- Number of cells that are plasmolysed which can then be used to calculate a percentage
- Control
- Volume of solution, we used 5cm3 pipettes each time
- Type of plant cell, each plant cell will have different default water concentrations and thickness of the membrane so it is important to use the same cell each time
Equipment
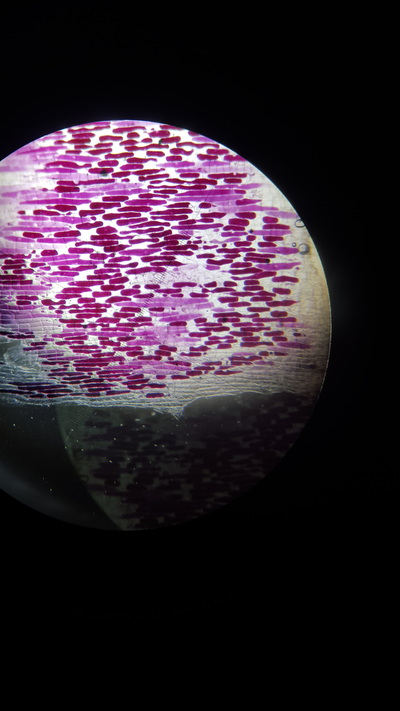
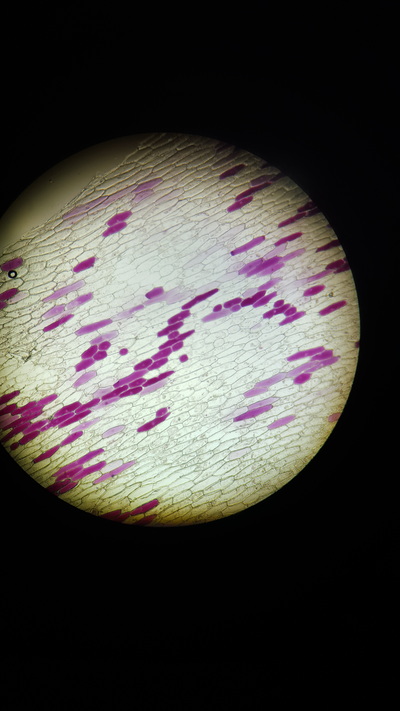
- Plant tissue with single cell layer ( a red onion skin cell)
- Six salt solutions of a suitable range for the tissue used
- We used 0.1 , 0.2, 0.3, 0.4, 0.5 and 0.6 mol dm-3 of salt solution
- Distilled water
- Watch glasses
- Measuring cylinders
- 5cm3 syringes
- Pipettes
- Filter paper
- Scalpel
- Labels or pen for labelling
- Microscope, slides and coverslips
- Iodine solution
Method
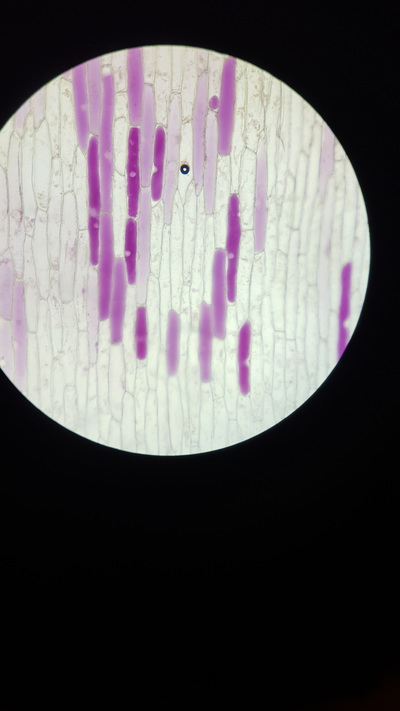
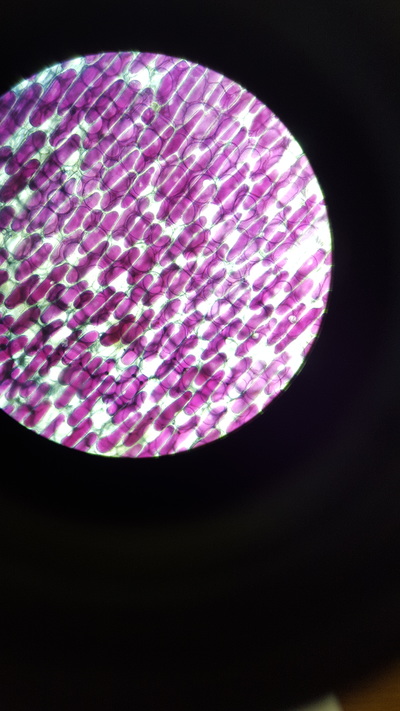
- Take a thin section of onion skin cell, this is because it is red and easily visible under a microscope
- Label watch glass with concentration of salt solution used, then put the onion cell in the watch glass and add the appropriate salt solution and leave for 20 minutes
- Remove tissue with forceps and place on a slide, again labelled with the correct concentration. Dry the solution with filter paper to suck up the water
- Place a coverslip on the onion making sure to remove any liquid
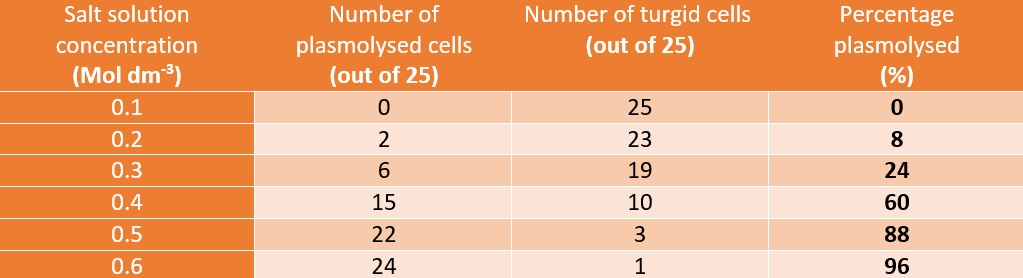
- Observe 25 cells and judge which have become plasmolysed. Calculate a percentage from this value
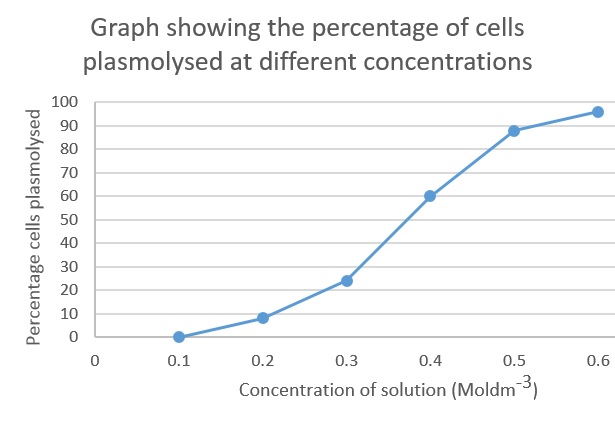
This graph shows there is a positive correlation between concentration of a salt solution and the percentage of cells that have plasmolysed. The solution was isotonic, relative to the onion cells at 0.1 Mol dm-3. This is because the cells did not change their structure meaning that the concentrations were similar. The solution from then on was hypotonic as water went from the cells to the solution (causing the cells to ‘shrivel’) showing it has a lower osmotic potential then the cells.